
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

Lupin Limited, a global pharmaceutical company, has officially received approval from the US FDA for its Abbreviated New Drug Application for Bromfenac Ophthalmic Solution, 0.09%. This approval allows Lupin to introduce a generic counterpart to Bausch & Lomb Inc.’s Bromday…

The Medicines and Healthcare products Regulatory Agency (MHRA) has recently issued a National Patient Safety Alert due to reports of increased intraocular pressure in patients recently implanted with EyeCee One preloaded and EyeCee One Crystal preloaded intraocular lenses (IOLs), which…

Caeregen Therapeutics announced that it has received a $1.4 million Phase 2 Small Business Innovation Research (SBIR) grant from the National Institutes of Health (NIH) and National Eye Institute (NEI). The grant has been awarded to further the advancement of…

After the initial approval of Apellis Pharmaceuticals’ geographic atrophy drug Syfovre (pegcetacoplan injection), six cases of retinal vasculitis have been reported. The ASRS Research and Safety in Therapeutics (ReST) Committee issued a notification disclosing these safety concerns to ASRS members,…
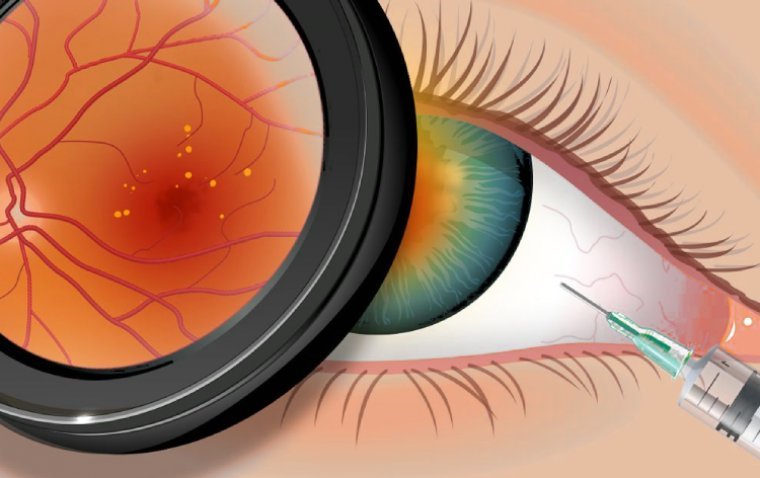
A recent study conducted by Columbia University researchers suggests that newly developed eye drops could potentially offer a more effective and comfortable treatment option for a common eye disease currently managed through eye injections. The eye disease in question is…

Cellusion, a Tokyo-based biotech company, announced the successful completion of its Series C Round financing, raising an impressive 2.83 billion yen, equivalent to $21 million. The primary focus of this funding is to accelerate the advancement of Cellusion’s lead program,…

Pharmedica USA has issued a worldwide recall for two lots of Purely Soothing, 15% MSM Drops, at the consumer level due to non-sterility concerns. The use of contaminated eye drops can pose a risk of eye infections and vision loss.…

Bausch + Lomb announced that the FDA has granted approval for Miebo (perfluorohexyloctane ophthalmic solution; previously referred to as NOV03) to treat the signs and symptoms of dry eye disease (DED). Miebo is the first and only FDA-approved treatment for…

Papilledema is the swelling of the optic nerve as it enters the back of the eye due to raised intracranial pressure. Fluid surrounding the brain is constantly produced and reabsorbed, maintaining just enough intracranial pressure to help protect the brain…

The colorful portion of the eye, the iris, is made up of a ring of muscle and connective tissue fibers that contract and stretch to open and close the pupil in response to the intensity of ambient light. The iris,…