
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

Eversight, a leading non-profit dedicated to the restoration of sight through the recovery, processing, and distribution of corneal tissue, announced a strategic partnership with Emmecell Biotech, a biotechnology company focused on developing novel cell therapies for ocular diseases. This collaboration…

Opthea Limited announced Dr. Arshad M. Khanani, MD, MA, FASRS, as its new Chief Medical Advisor. Dr. Khanani, renowned in retina specialization and clinical research, brings a wealth of expertise from his tenure at Sierra Eye Associates and his academic…

Opthea has reached a significant milestone in its quest to combat wet age-related macular degeneration (AMD) with the completion of patient enrollment in the COAST phase 3 pivotal clinical trial. The trial investigates sozinibercept (OPT-302), a promising vascular endothelial growth…
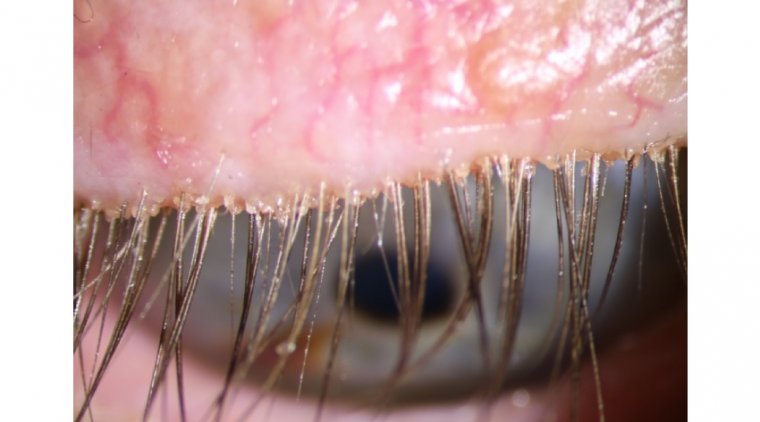
A team of investigators, led by Elizabeth Yeu, MD, from Virginia Eye Consultants in Norfolk, Virginia, reported the safety and efficacy of lotilaner ophthalmic solution, 0.25% (TP-03, Tarsus Pharmaceuticals Inc.), in treating Demodex blepharitis. Demodex mites are the most common…

Swiss-based pharmaceutical company Roche has announced the discontinuation of its oral diabetic retinopathy treatment, vicasinabin, after successfully completing Phase II clinical trials. In a recent presentation to investors, Roche confirmed its decision to halt further investment in vicasinabin, a promising…

EyePoint Pharmaceuticals announced the dosing of the first patient in the VERONA clinical trial for their investigational therapy, EYP-1901, for the treatment of Diabetic Macular Edema (DME). EYP-1901, a sustained delivery therapy, incorporates vorolanib, a selective tyrosine kinase inhibitor, formulated…

Eyenovia announced the successful completion of the FDA inspection for its facility in Redwood City, California, marking a significant milestone in the company’s manufacturing capabilities. This achievement signals the green light for the facility to engage in final assembly, packaging,…

Okyo Pharma’s first investigational new drug (IND) application for OK-101 has been cleared by the U.S. Food and Drug Administration (FDA), marking a pivotal moment in the development of potential therapies for neuropathic corneal pain. Initially proposed as an open-label…

According to the National Eye Institute (NEI), a trial conducted by the Pediatric Eye Disease Investigator Group (PEDIG) and funded by the NEI revealed that the use of low-dose atropine eye drops (concentration 0.01%) did not demonstrate superiority over a…

Glaukos has received FDA approval for iDose TR (travoprost intracameral implant 75 mcg), a groundbreaking prostaglandin analog intracameral implant designed to reduce intraocular pressure (IOP) in individuals with ocular hypertension or open-angle glaucoma. The iDose TR procedure, sanctioned for a…