
Newsletter Subscribe
Enter your email address below and subscribe to our newsletter


Enter your email address below and subscribe to our newsletter

In a recent announcement, Ocuphire Pharma disclosed that it has secured approval from the U.S. Food and Drug Administration (FDA) under a special protocol assessment (SPA) for the clinical trial protocol and planned statistical analysis of its LYNX-2 phase 3…

EyeBio announced the successful completion of an extension to its Series A financing, resulting in a total raised amount of $130 million to date. The funding will be utilized by EyeBio to accelerate the company’s clinical development program and expand…

VSY Biotechnology GmbH is proud to announce the groundbreaking development of Varliciment, a revolutionary anti-VEGF agent for the treatment of age-related macular degeneration (AMD). Varliciment distinguishes itself from existing anti-VEGF agents like brolucizumab, namely Novartis’ Beovu, by being non-generic. This…

Ocular Therapeutix has obtained written FDA approval for the comprehensive design of its pivotal phase 3 clinical trial, known as the SOL trial, which evaluates Axpaxli (axitinib intravitreal implant) for treating wet age-related macular degeneration (AMD) under a Special Protocol…

OnPoint Vision has officially received FDA approval for their investigational device exemption (IDE) application, initiating the phase 1 in the pivotal clinical trial for the AccuraSee Intraocular Pseudophakic Capsular Lens (IOPCL) Magnifier (MAG). This trial focuses on the secondary implantation…

Ocuphire Pharma and Viatris announced that the FDA has granted approval for Ryzumvi (phentolamine ophthalmic solution) 0.75%. This approval specifically authorizes its use in the treatment of pharmacologically-induced mydriasis, produced by adrenergic agonists (such as phenylephrine) or parasympatholytic agents (like…

Neurophth Therapeutics announced that the Australian Therapeutic Goods Administration (TGA) has granted registration and approval for its candidate drug, NFS-05, for clinical trials aimed at addressing autosomal dominant optic atrophy (ADOA). According to a company news release, ADOA is an…
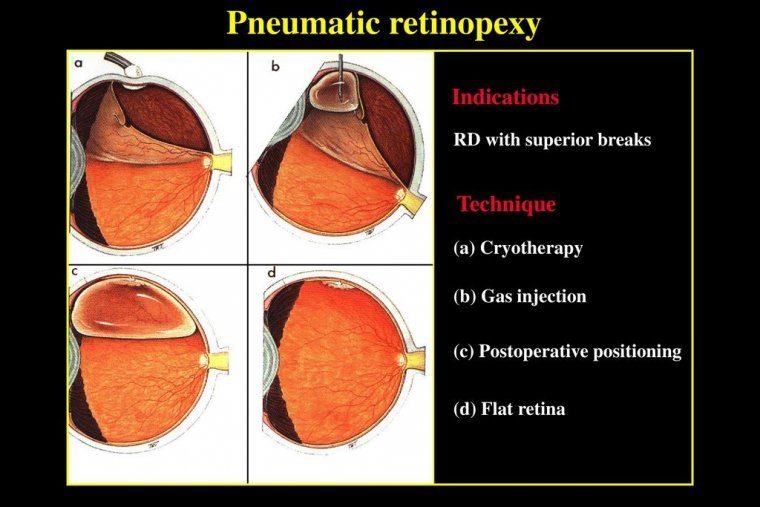
Pneumatic retinopexy is an in-office procedure used to repair certain types of retinal detachments. Pneumatic retinopexy typically treats rhegmatous retinal detachments. The eye is numbed with anesthesia so there is no pain. A gas bubble is injected into the eye…

Development of active glasses for mitigating presbyopia has become an active area of research, facilitated by the miniaturization of focus-tunable lenses as well as the increased processing power of modern computers. Alcon the global leader in eye care dedicated to…

Marinomed Biotech has announced the introduction of a new Carragelose eye drop product, known as MAM-1001-3, to their existing Carragelose OTC portfolio. The product has obtained certification under the Medical Device Directive (MDD) and is currently undergoing preparations for marketing.…